
Jan . 30, 2026 14:40 Back to list
A Comprehensive Guide to Make Glacial Acetic Acid and Its Industrial Applications
Make Glacial Acetic Acid: A Comprehensive Guide
Glacial acetic acid, a highly concentrated form of acetic acid, is a crucial chemical compound used in various industries. Understanding its production, applications, and safety considerations is vital. This article provides a detailed exploration of how to make glacial acetic acid, covering different methods, quality control, and sourcing from a reputable supplier like YSXL Glacial Acetic Acid. We’ll delve into the intricacies of this important chemical and its role in diverse applications.

Understanding Glacial Acetic Acid: Properties and Uses
Glacial acetic acid (CH₃COOH) is so named because it freezes to a crystalline solid resembling ice at approximately 16.7°C (62°F). It’s a colorless liquid with a pungent, vinegary odor. Its primary use is as a chemical reagent in the production of various compounds including vinyl acetate monomer (VAM), acetic anhydride, and esters. It’s also utilized in the food industry as a preservative and flavoring agent, in the pharmaceutical industry, and in textile processing. Its high acidity makes it an effective disinfectant and cleaning agent. Sourcing high-quality glacial acetic acid from a reliable supplier like YSXL Glacial Acetic Acid ensures optimal performance in these diverse applications.
Methods to Make Glacial Acetic Acid
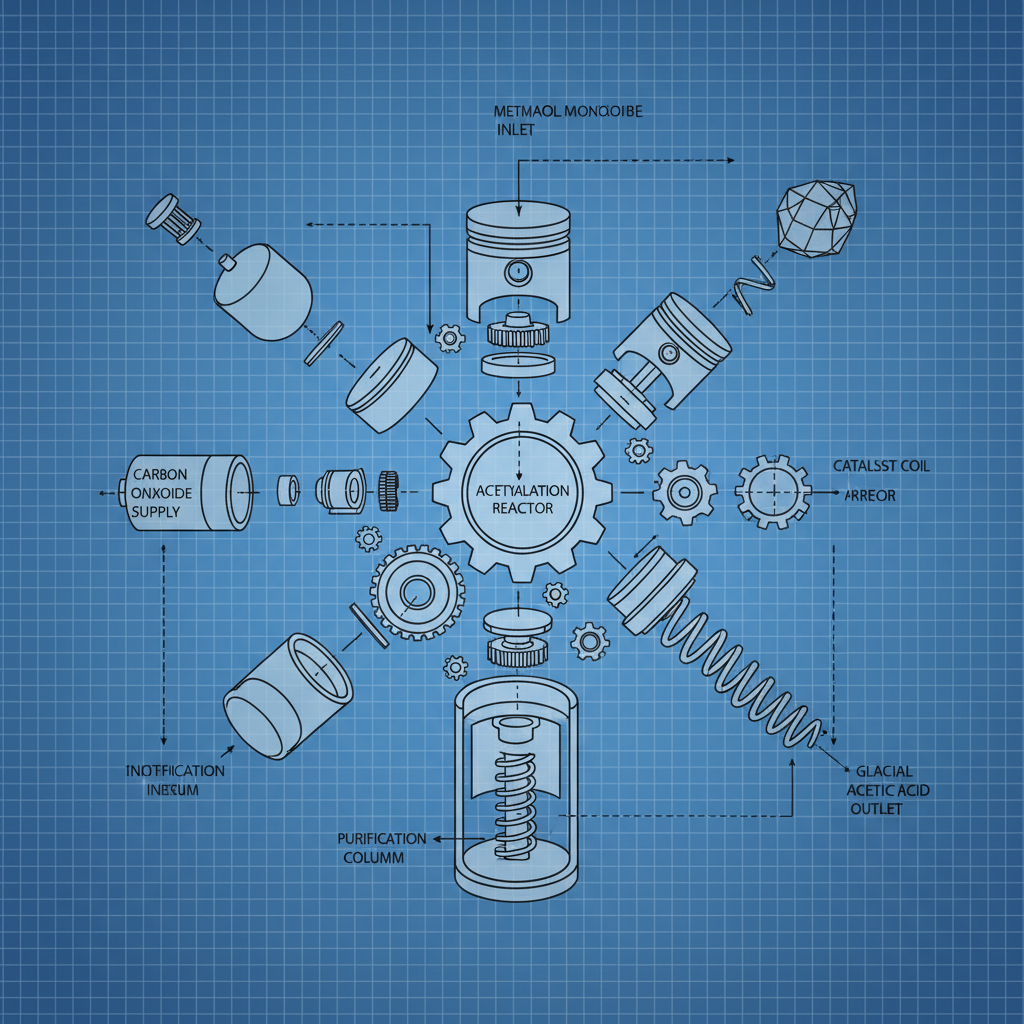
While industrial production is complex, understanding the core principles behind making glacial acetic acid is beneficial. Historically, acetic acid was produced through the fermentation of ethanol by Acetobacter bacteria. However, modern industrial processes primarily rely on the carbonylation of methanol. This involves reacting methanol with carbon monoxide in the presence of a catalyst (typically rhodium or iridium complexes). The resulting product is then purified through distillation to achieve the high concentration necessary for glacial acetic acid. Another method is the oxidation of acetaldehyde, but this is less common. It's crucial to note that attempting to replicate these industrial processes without appropriate expertise and equipment is extremely dangerous.
Key Methods: Carbonylation of Methanol, Fermentation of Ethanol, Oxidation of Acetaldehyde. Industrial production requires specialized equipment and safety measures.
Carbonylation of Methanol: The Industrial Standard
The carbonylation of methanol is the dominant method for glacial acetic acid production due to its efficiency and high yield. The process occurs in the liquid phase using a homogeneous catalyst, typically a rhodium-based complex. The reaction is carried out under pressure and controlled temperature conditions. The resulting product stream contains acetic acid, water, and unreacted methanol. Purification is then achieved through fractional distillation, leveraging the different boiling points of the components. This process demands rigorous safety protocols and precise control of process parameters to ensure both product quality and operational safety.
Carbonylation Process Highlights:
• High efficiency and yield
• Utilizes rhodium or iridium catalysts
• Requires precise control of temperature and pressure
• Relies on fractional distillation for purification
Glacial Acetic Acid: Product Specifications & Quality Control
Maintaining the high purity of glacial acetic acid is paramount. Reputable suppliers like YSXL Glacial Acetic Acid adhere to stringent quality control measures. Key specifications typically include:
Safety Considerations When Handling Glacial Acetic Acid
Glacial acetic acid is highly corrosive and poses significant safety hazards. Direct contact can cause severe burns to skin and eyes. Inhalation of vapors can irritate the respiratory system. Always wear appropriate personal protective equipment (PPE), including gloves, eye protection, and a respirator when handling this chemical. Work in a well-ventilated area. In case of contact, immediately flush affected areas with copious amounts of water and seek medical attention. Store glacial acetic acid in a cool, dry, and well-ventilated area, away from incompatible materials. Always consult the Safety Data Sheet (SDS) for detailed safety information before handling.
Conclusion: Sourcing Quality Glacial Acetic Acid
Obtaining high-quality glacial acetic acid is crucial for successful applications. YSXL Glacial Acetic Acid provides consistently pure and reliable products, backed by rigorous quality control and a commitment to safety. By partnering with a trusted supplier, you can ensure the integrity of your processes and the safety of your operations.
Frequently Asked Questions (FAQs)
What are the primary applications of glacial acetic acid?
Glacial acetic acid is a versatile chemical used extensively in the production of vinyl acetate monomer (VAM), a key component in paints, adhesives, and textiles. It also serves as a solvent in chemical synthesis, a preservative in food, and a reagent in pharmaceutical manufacturing. Furthermore, it finds applications in the production of cellulose acetate, used in photographic film and cigarette filters. Its highly acidic nature makes it an effective cleaning and disinfecting agent, and it's used in various industrial processes.
How should I store glacial acetic acid safely?
Glacial acetic acid should be stored in tightly closed, corrosion-resistant containers, in a cool, dry, and well-ventilated area. It's crucial to keep it away from heat, sparks, open flames, and incompatible materials such as strong bases, oxidizing agents, and metals. Avoid storing it in direct sunlight. The storage area should be equipped with appropriate spill containment measures. Always refer to the Safety Data Sheet (SDS) for specific storage recommendations.
What PPE is essential when handling glacial acetic acid?
Due to its corrosive nature, handling glacial acetic acid requires comprehensive personal protective equipment (PPE). This includes chemical-resistant gloves (such as nitrile or neoprene), a face shield or chemical splash goggles, and a chemical-resistant apron or suit. If there’s a risk of vapor inhalation, a respirator with an acid gas filter should be used. It's essential to ensure the PPE is in good condition and fits properly.
What should I do in case of a glacial acetic acid spill?
In the event of a glacial acetic acid spill, immediately evacuate the area and ensure proper ventilation. Wear appropriate PPE before attempting to clean up the spill. Neutralize the acid with a suitable absorbent material, such as sodium bicarbonate or a commercial acid spill kit. Avoid flushing the spill down the drain. Dispose of the contaminated absorbent material according to local regulations. Report the spill to the appropriate authorities if required.
-
Industrial Applications and Properties of 75 acetic acid Solution
NewsApr.16,2026
-
Properties and Industrial Applications of 64 19 7 acetic acid
NewsApr.09,2026
-
Comprehensive Guide to 56 Acetic Acid Applications and Reliable Sourcing
NewsApr.07,2026
-
Comprehensive Guide to 5 Glacial Acetic Acid for International Trade and Industrial Applications
NewsApr.04,2026
-
Exploring 100 Acetic Acid Properties Applications and Reliable Sourcing
NewsMar.31,2026
-
Comprehensive Guide to 10 Glacial Acetic Acid Properties and Applications
NewsMar.28,2026
