
Feb . 11, 2026 02:15 Back to list
Learn How to Make Glacial Acetic Acid from Vinegar Safely and Effectively
Making Glacial Acetic Acid from Vinegar: A Comprehensive Guide
Glacial acetic acid, a highly concentrated form of acetic acid, is a vital chemical in various industries, from food processing to pharmaceuticals. While commercially produced, it's possible to create it from common vinegar through a process of distillation and concentration. This article details the methods, safety precautions, and considerations involved in making glacial acetic acid from vinegar, providing a clear understanding for those interested in this chemical process. We'll explore the equipment, techniques, and critical safety measures required for successful and safe production.
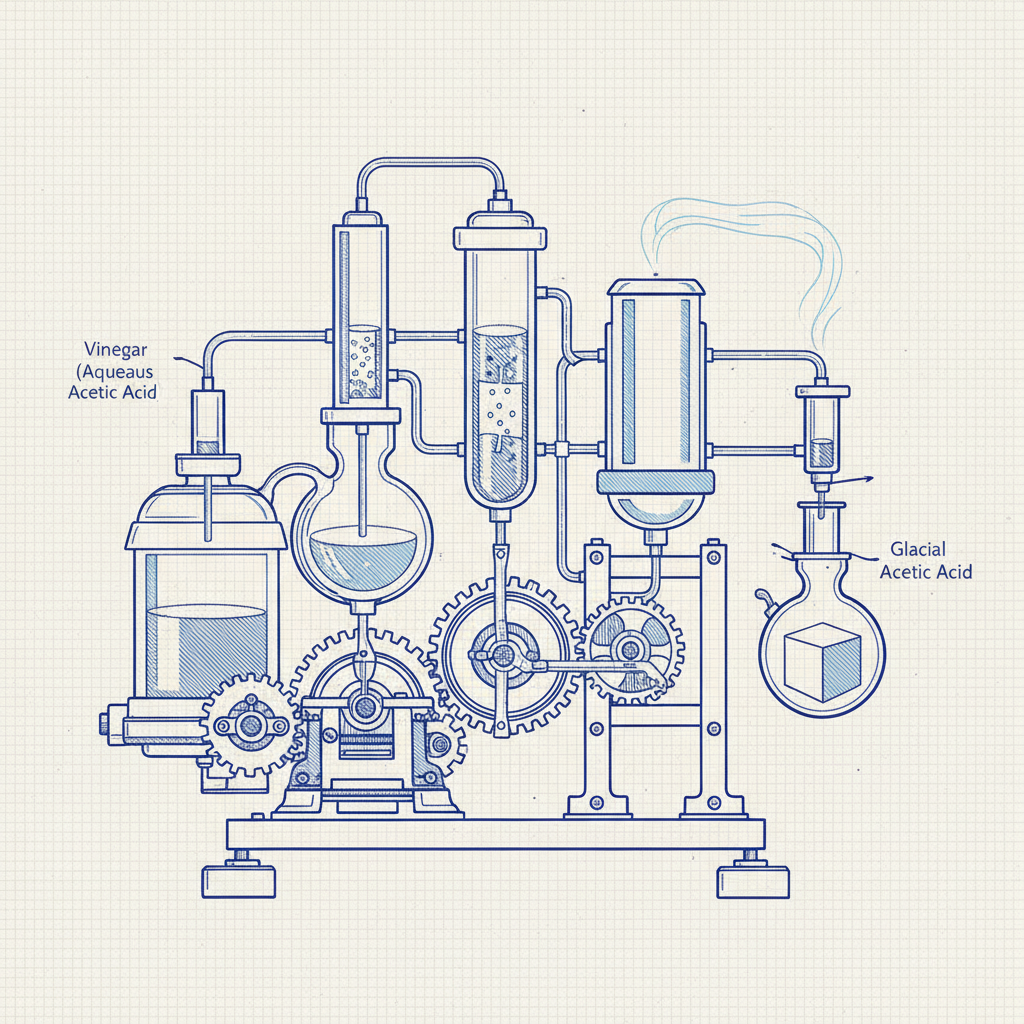
Understanding the Chemistry: From Vinegar to Glacial Acetic Acid
Vinegar is a dilute solution of acetic acid (typically 5-8% concentration) in water. Glacial acetic acid, on the other hand, is nearly pure acetic acid (99-100%). The conversion involves increasing the concentration of acetic acid by removing water through distillation. This process relies on the differing boiling points of water (100°C / 212°F) and acetic acid (118°C / 244°F). Careful control of temperature is crucial to effectively separate the acetic acid from the water. It’s important to remember that the process doesn’t create acetic acid; it simply concentrates the existing acid in the vinegar. YSXL Glacial Acetic Acid offers a consistent, high-quality product for various applications.
Key Point: Distillation leverages the difference in boiling points to separate liquids. Controlling temperature is vital for efficient separation and preventing unwanted mixtures.
Essential Equipment for Distillation
Successfully making glacial acetic acid from vinegar requires specific equipment. A distillation apparatus is central to the process. This typically includes a heat source (hot plate or heating mantle), a distillation flask, a condenser (water-cooled), and a receiving flask. A thermometer is essential for monitoring temperature during distillation. High-quality glassware is recommended to withstand the corrosive nature of acetic acid. Safety equipment, including gloves, eye protection, and a well-ventilated workspace, is also paramount. Consider a fractional distillation setup for improved purity.
Essential Equipment Checklist:
• Distillation Flask
• Condenser (Water-Cooled)
• Receiving Flask
• Heat Source (Hot Plate/Mantle)
• Thermometer
• Safety Gear (Gloves, Goggles)
The Distillation Process: Step-by-Step Guide
The distillation process begins by carefully heating the vinegar in the distillation flask. Monitor the temperature closely. As the mixture heats, the water will begin to vaporize first. The vapor is then passed through the condenser, where it is cooled back into liquid form and collected in the receiving flask. The goal is to collect the acetic acid vapor after the water has largely evaporated, maintaining a temperature just below the boiling point of acetic acid. This is often done slowly, collecting fractions and discarding the initial portions, which contain a higher water content. This process requires patience and precision.
Safety Precautions When Making Glacial Acetic Acid
Working with acetic acid, especially in concentrated form, requires strict adherence to safety protocols. Acetic acid is corrosive and can cause severe burns upon contact with skin or eyes. Always wear appropriate personal protective equipment (PPE), including gloves, safety goggles, and a lab coat. Work in a well-ventilated area to avoid inhaling the fumes. Avoid open flames near the distillation apparatus. Have a neutralizing agent, such as baking soda, readily available in case of spills. Purchasing glacial acetic acid from a reputable supplier like YSXL is a safer alternative to home production.
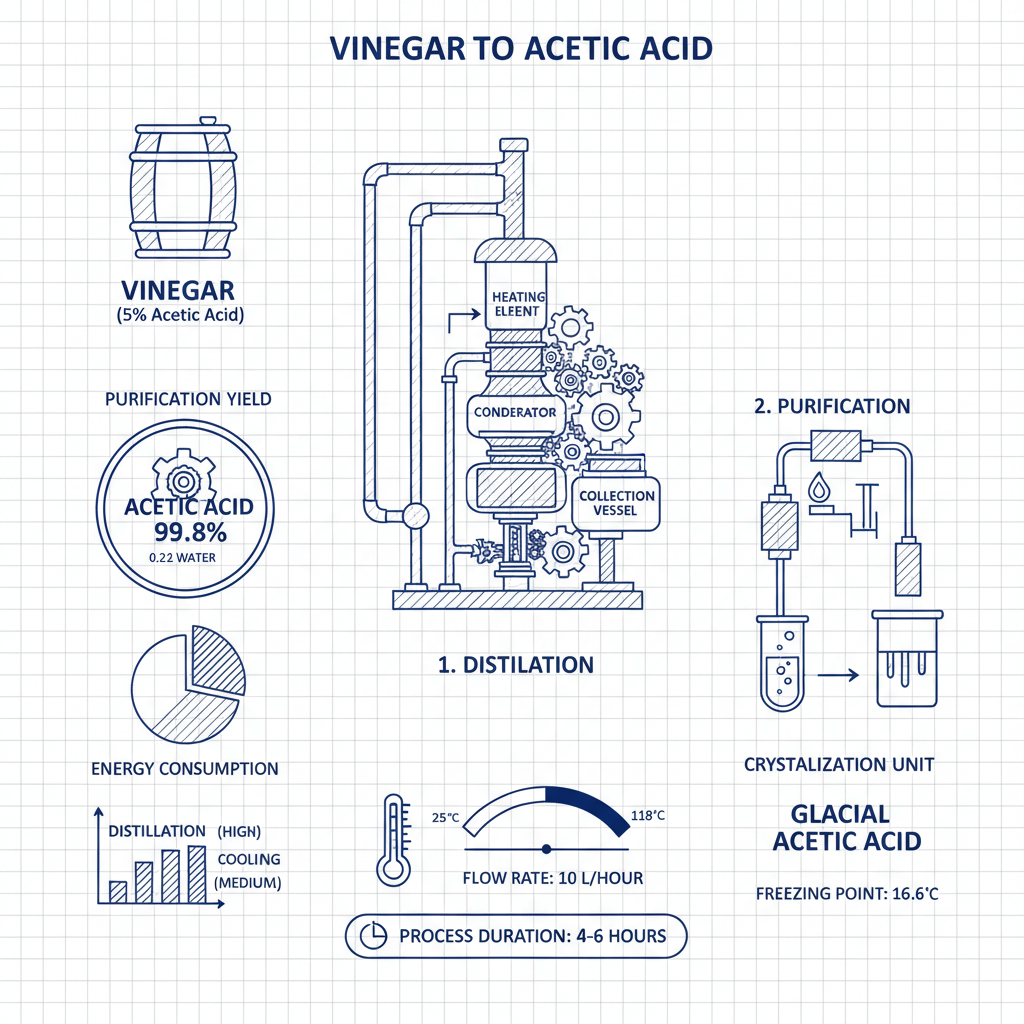
Purity and Considerations: Why Commercial Glacial Acetic Acid is Often Preferred
Achieving truly glacial purity (99-100%) through home distillation is challenging. Impurities from the vinegar and incomplete separation can remain in the final product. Commercially produced glacial acetic acid undergoes rigorous purification processes to ensure consistent quality and purity. This is crucial for applications requiring high precision, such as laboratory experiments or pharmaceutical manufacturing. While distillation is a valuable learning exercise, sourcing glacial acetic acid from YSXL guarantees a reliable, high-quality product tailored to your specific needs.
Conclusion: Weighing the Options
While it’s possible to make glacial acetic acid from vinegar, it requires specialized equipment, careful technique, and a strong emphasis on safety. The resulting purity may not be sufficient for demanding applications. Considering these factors, purchasing high-quality glacial acetic acid from a trusted supplier like YSXL Glacial Acetic Acid often proves to be a more practical and safe solution.
Frequently Asked Questions (FAQs)
What type of vinegar is best suited for this process?
White distilled vinegar is generally preferred because it's the purest form of vinegar available, containing minimal impurities that could interfere with the distillation process. Other types of vinegar, such as apple cider vinegar, contain additional compounds that can affect the purity of the final product. The higher the initial acetic acid concentration in the vinegar, the less volume you'll need to distill to obtain a significant amount of glacial acetic acid. It's important to note that even with white distilled vinegar, achieving 100% purity through distillation can be challenging.
What are the potential dangers of improper distillation?
Improper distillation can lead to several dangers. Firstly, acetic acid is corrosive and can cause severe burns if it comes into contact with skin or eyes. Secondly, flammable vapors can accumulate, creating a fire hazard. Lastly, incomplete separation of water from acetic acid can result in a product with reduced purity and potentially undesirable properties. Ensuring adequate ventilation, wearing appropriate PPE, and carefully controlling the temperature are crucial safety measures.
Is it legal to distill acetic acid at home?
The legality of distilling acetic acid at home varies depending on your location. Some jurisdictions may have regulations regarding the production of chemicals, even for personal use. It is essential to check local laws and regulations before attempting to distill acetic acid. Additionally, it's crucial to ensure that the process is conducted safely and responsibly to avoid any legal repercussions. Purchasing from a reputable supplier like YSXL bypasses any legal concerns.
What are the common uses of glacial acetic acid?
Glacial acetic acid has a wide range of applications, including as a reagent in chemical synthesis, a solvent, a food preservative, and in the production of cellulose acetate (used in textiles and plastics). It's also used in the pharmaceutical industry, in the production of various chemical compounds, and as an analytical reagent. Due to its high concentration, it's more effective in these applications compared to diluted vinegar.
-
Industrial Applications and Properties of 75 acetic acid Solution
NewsApr.16,2026
-
Properties and Industrial Applications of 64 19 7 acetic acid
NewsApr.09,2026
-
Comprehensive Guide to 56 Acetic Acid Applications and Reliable Sourcing
NewsApr.07,2026
-
Comprehensive Guide to 5 Glacial Acetic Acid for International Trade and Industrial Applications
NewsApr.04,2026
-
Exploring 100 Acetic Acid Properties Applications and Reliable Sourcing
NewsMar.31,2026
-
Comprehensive Guide to 10 Glacial Acetic Acid Properties and Applications
NewsMar.28,2026
