
56 acetic acid, also known as glacial acetic acid, is a fundamental chemical compound with widespread applications across diverse industries. Its significance stems from its properties as a versatile solvent, a crucial reagent in chemical synthesis, and a key component in various manufacturing processes. Understanding 56 acetic acid is therefore critical for professionals in chemistry, pharmaceuticals, textiles, food production, and many other fields.
Globally, the demand for 56 acetic acid is consistently high, driven by economic growth and industrial expansion, particularly in emerging markets. According to industry reports, the global acetic acid market is projected to reach substantial figures in the coming years, showcasing its enduring importance. This widespread demand presents both opportunities and challenges for manufacturers and distributors, requiring efficient supply chains and sustainable production practices.
Beyond its industrial uses, 56 acetic acid plays a crucial role in scientific research and development, serving as a vital tool in numerous experiments and analytical procedures. Its relatively low cost and ready availability further contribute to its widespread adoption. The ability to easily procure and utilize 56 acetic acid makes it a cornerstone of many laboratory processes, enabling scientific advancement across various disciplines.
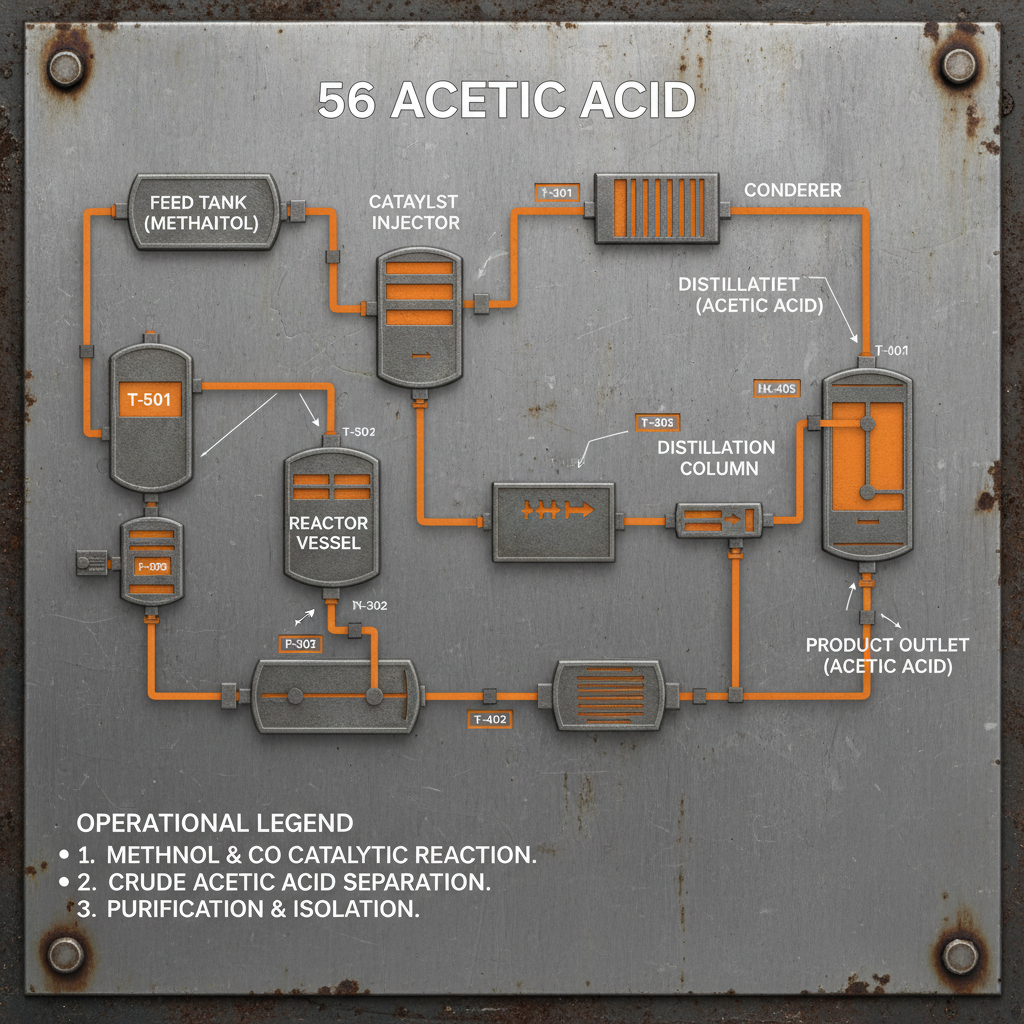

56 acetic acid is characterized by its distinct pungent odor and colorless liquid state. Its chemical formula, CH3COOH, reveals its simple molecular structure, yet this belies its powerful reactivity. Understanding these foundational properties is vital for safe handling and effective application.
The high solubility of 56 acetic acid in water, as well as its ability to dissolve numerous organic compounds, makes it an exceptional solvent in a multitude of processes. This solubility stems from its polar nature and its capacity to form hydrogen bonds, contributing to its versatility in various industrial applications.
The production of 56 acetic acid is concentrated in several key regions globally, with significant output from China, the United States, and Europe. These regions boast established chemical manufacturing infrastructure and benefit from access to raw materials like methanol and ethylene. Effective supply chain management is therefore crucial for meeting global demand.
The supply chain for 56 acetic acid is often complex, involving multiple stakeholders from raw material suppliers to manufacturers, distributors, and end-users. Disruptions in any part of this chain, such as geopolitical events or logistical challenges, can impact availability and pricing.
Increasingly, there's a focus on regionalizing production to reduce reliance on long-distance supply routes and enhance resilience. This trend aims to address potential vulnerabilities and ensure a more stable supply of 56 acetic acid to local markets.
56 acetic acid is a weak carboxylic acid, meaning it doesn't fully dissociate in solution. This characteristic is fundamental to its reactivity and its ability to participate in various chemical reactions, including esterification, neutralization, and oxidation. Its relatively low pKa value of around 4.76 indicates its acidity.
A key chemical characteristic of 56 acetic acid is its ability to form azeotropes with water. This means that mixtures of 56 acetic acid and water boil at a constant temperature and composition, impacting purification processes. Understanding azeotrope formation is crucial for achieving high purity in industrial applications.
The reactivity of 56 acetic acid stems from its carboxyl group (-COOH). This group allows it to readily participate in reactions forming esters, amides, and other important chemical compounds. These derivatives find widespread uses in flavors, fragrances, and polymers.
Assessing the performance of 56 acetic acid in various applications requires considering several key metrics. Purity is paramount, with specifications often exceeding 99.8% for sensitive applications like pharmaceutical synthesis and analytical chemistry.
Another crucial performance metric is color, as discoloration can indicate the presence of impurities or degradation products. Color is typically measured using the APHA scale, with lower values indicating higher quality. Understanding these metrics allows for optimal control and consistency.
The demand for 56 acetic acid varies significantly across different regions, reflecting diverse economic structures and industrial activities. Asia-Pacific represents the largest consumer, driven by its rapidly expanding textile and chemical industries.
North America and Europe exhibit robust demand, primarily from the pharmaceutical, food, and chemical sectors. Regulatory standards and a focus on high-quality products influence demand patterns in these regions.
The production of 56 acetic acid can have environmental impacts, including greenhouse gas emissions and waste generation. However, advancements in production technologies are mitigating these effects through energy efficiency improvements and waste reduction strategies.
Increasingly, bio-based production methods utilizing renewable feedstocks are gaining traction. These methods offer a more sustainable alternative to traditional fossil fuel-based processes, reducing the carbon footprint of 56 acetic acid production.
Responsible handling and disposal practices are crucial for minimizing the environmental impact of 56 acetic acid throughout its lifecycle. Adhering to environmental regulations and implementing effective waste management systems are essential for promoting sustainability.
Future trends in the 56 acetic acid industry are focused on sustainable production, process intensification, and the development of novel applications. Research and development efforts are concentrated on optimizing existing production methods and exploring alternative feedstocks.
The integration of digital technologies, such as process monitoring and control systems, is enhancing efficiency and reducing waste. These technologies enable real-time optimization and improved quality control throughout the production process.
Emerging applications for 56 acetic acid include its use in the production of biodegradable plastics and as a solvent in green chemistry processes. These innovations promise to further expand the versatility and sustainability of 56 acetic acid.
| Raw Material Cost | Process Technology | Energy Consumption | Waste Management Efficiency |
|---|---|---|---|
| Low | Advanced Catalysis | Optimal | High |
| Medium | Conventional Oxidation | Moderate | Medium |
| High | Carbonylation | High | Low |
| Medium | Methanol Carbonylation | Moderate | Medium |
| Low | Bio-based Fermentation | Low | High |
| Medium | Ethylene Oxidation | Moderate | Medium |
56 acetic acid serves as a crucial solvent in the production of various chemicals, including plastics, synthetic fibers, and pharmaceuticals. It is also vital in the manufacturing of vinyl acetate monomer (VAM), a key component in paints, adhesives, and textiles. Additionally, it finds use in food processing as a preservative and flavoring agent.
Due to its corrosive nature, 56 acetic acid requires careful handling. Personal protective equipment (PPE), including gloves, goggles, and a lab coat, should always be worn. Work should be conducted in a well-ventilated area to avoid inhalation of vapors. In case of skin contact, immediately flush with copious amounts of water and seek medical attention if irritation persists.
The purity of 56 acetic acid is commonly determined through gas chromatography (GC), which separates and quantifies different components in the sample. Titration with a standardized base is also used to determine the acid content. Colorimetric analysis, using the APHA scale, assesses the presence of impurities causing discoloration.
Traditional 56 acetic acid production can contribute to greenhouse gas emissions and generate waste streams. Sustainable practices, such as utilizing bio-based feedstocks and implementing efficient waste management systems, are crucial for minimizing environmental impact. Efforts are underway to develop cleaner production technologies.
Temperature significantly impacts the properties of 56 acetic acid. As temperature increases, its viscosity decreases, and its vapor pressure rises. Its density also changes with temperature, and its reactivity can be influenced, particularly in reactions involving catalysts. Understanding these temperature-dependent properties is critical for process control.
Emerging trends include the development of more efficient catalysts for methanol carbonylation, the utilization of carbon dioxide as a feedstock, and the implementation of advanced process control systems based on machine learning and artificial intelligence. These technologies aim to enhance sustainability, reduce costs, and improve product quality.
In conclusion, 56 acetic acid remains a vital chemical compound with widespread applications and a significant global presence. Its versatility as a solvent, reagent, and intermediate, coupled with ongoing innovation in production technologies, ensures its continued importance across numerous industries. Understanding its properties, applications, and environmental implications is critical for professionals and researchers alike.
Looking ahead, the future of 56 acetic acid lies in the pursuit of sustainable production methods, the development of novel applications, and the integration of digital technologies. By embracing these advancements, we can unlock the full potential of 56 acetic acid while minimizing its environmental impact and maximizing its value to society. For more information and to explore our product offerings, please visit our website: www.ysxlglacialaceticacid.com.