In the complex landscape of food science and industrial chemistry, glacial ch3cooh stands as a cornerstone reagent, essential for maintaining acidity, enhancing preservation, and facilitating countless chemical syntheses. Its purity and concentrated nature make it an indispensable tool for manufacturers who require precise control over pH levels to ensure product stability and safety.
Globally, the demand for high-purity acetic acid derivatives has surged as the food and pharmaceutical industries shift toward more stringent quality standards. Understanding the nuances of glacial ch3cooh allows producers to optimize their formulation processes, reduce waste, and meet the evolving regulatory requirements of international markets such as the FDA and EFSA.
By exploring the chemical properties, industrial applications, and safety protocols associated with this potent compound, businesses can leverage its versatility to create superior food-grade additives. This guide provides a comprehensive analysis of how glacial ch3cooh drives innovation in food manufacturing and sustainable chemical production.


The global market for glacial ch3cooh is intrinsically linked to the growth of the vinyl acetate monomer (VAM) and purified terephthalic acid (PTA) sectors. According to recent industrial reports, the scale of acetic acid production has expanded significantly to keep pace with the burgeoning demand for biodegradable plastics and food-grade preservatives, reflecting a global shift toward safer chemical additives.
However, the industry faces a recurring challenge: balancing the extreme purity required for food-grade applications with the cost-efficiencies of mass production. Many manufacturers struggle with residue contaminants that can alter the flavor profile of food products, making the sourcing of ultra-pure glacial ch3cooh a critical strategic priority for quality assurance teams worldwide.
In simple chemical terms, glacial ch3cooh refers to anhydrous acetic acid, meaning it is nearly 100% pure and water-free. The term "glacial" is derived from the substance's tendency to freeze into ice-like crystals just below room temperature (approximately 16.6°C), a physical property that signifies its high concentration and potency.
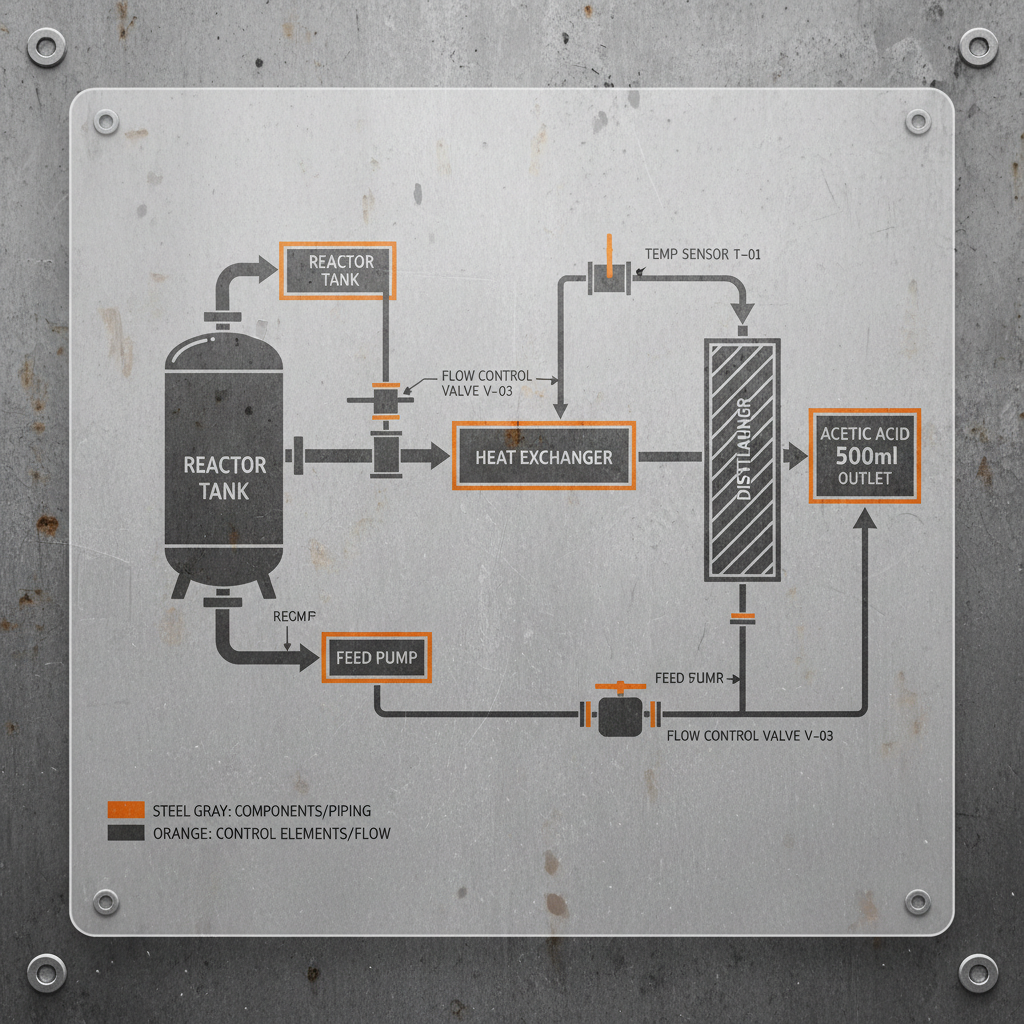
Within the context of modern industry, this compound serves as more than just a solvent; it is a vital building block for organic synthesis. In the food additive sector, it is the precursor to various acetates and is used directly as a pH regulator to inhibit the growth of spoilage microorganisms, thereby extending the shelf life of processed foods without compromising taste.
The humanitarian and societal relevance of glacial ch3cooh is seen in its role in food security. By enabling more efficient preservation methods, it helps reduce global food waste and ensures that nutritious products can be transported over long distances to remote regions without spoiling, aligning with global sustainability goals.
The effectiveness of glacial ch3cooh in industrial settings is determined primarily by its chemical purity. Impurities such as heavy metals or residual catalysts from the methanol carbonylation process can cause unwanted reactions in sensitive food formulations, making the "food grade" certification the gold standard for compliance.
Another critical factor is stability and scalability. Because glacial ch3cooh is highly corrosive, the materials used for storage and transport—such as high-grade stainless steel or specialized polymers—are essential components of the supply chain to prevent contamination and ensure operational safety.
Finally, cost-efficiency is achieved through precise dosage and concentration. By utilizing the concentrated form of glacial ch3cooh, manufacturers reduce shipping costs associated with water weight and lower their overall carbon footprint, provided they have the technical infrastructure to dilute it safely on-site.
The application of glacial ch3cooh spans across diverse food categories. In the pickling and canning industries, it is used to create the acidic environment necessary to prevent botulism and other bacterial contaminations. Beyond preservation, it acts as a flavor enhancer in condiments, providing the sharp, tangy profile expected in high-quality vinegars and sauces.
In pharmaceutical-grade food additives, glacial ch3cooh is employed in the production of various esters and salts that stabilize vitamins and minerals. Its role as a solvent in the extraction of natural flavors and essences further demonstrates its versatility in creating clean-label ingredients for the health-conscious consumer.
The long-term value of integrating high-quality glacial ch3cooh into a production line lies in its reliability. By ensuring consistent pH levels, manufacturers can reduce the rate of product spoilage and recalls, which directly protects the brand's reputation and enhances consumer trust in the safety of the food supply chain.
From a sustainability perspective, the transition toward bio-based glacial ch3cooh is creating a positive environmental impact. This innovation reduces dependence on fossil-fuel-derived methanol, lowering the carbon footprint of the food additive industry and promoting a circular economy where agricultural waste is converted into high-value chemical reagents.
The future of glacial ch3cooh is being shaped by the digital transformation of chemical plants. "Industry 4.0" is introducing AI-driven monitoring systems that can predict purity fluctuations in real-time, allowing for automated adjustments in the synthesis process to maintain absolute food-grade standards.
Furthermore, we are seeing a rise in the development of "green catalysts" that make the production of glacial ch3cooh more energy-efficient. These catalysts operate at lower temperatures and pressures, significantly reducing the energy intensity of the manufacturing process and aligning with global net-zero emission targets.
Another emerging trend is the integration of blockchain for traceability. By tagging batches of glacial ch3cooh from the source to the end food product, manufacturers can provide transparent proof of origin and purity, satisfying the demands of increasingly scrutinized global food safety regulations.
Despite its benefits, the handling of glacial ch3cooh presents significant logistical challenges, primarily due to its corrosive nature. Many facilities struggle with the initial capital expenditure required to install corrosion-resistant piping and automated dosing systems that prevent human exposure to concentrated fumes.
To overcome these hurdles, experts recommend a phased implementation of safety protocols and the use of modular dilution units. By automating the mixing process, companies can maintain the high purity of glacial ch3cooh while eliminating the risks associated with manual handling, thereby improving worker safety and operational efficiency.
Additionally, the challenge of regulatory variance between countries can be mitigated by adhering to the most stringent international standard (such as the FCC - Food Chemicals Codex). This ensures that products using glacial ch3cooh are compliant in any market, simplifying the export process and reducing legal risks for global food brands.
| Implementation Factor | Risk Level (1-10) | Mitigation Strategy | Efficiency Gain |
|---|---|---|---|
| Material Corrosion | 9 | Use 316L Stainless Steel | High |
| Purity Fluctuations | 4 | Real-time HPLC Monitoring | Medium |
| Worker Exposure | 8 | Closed-loop Dosing Systems | High |
| Regulatory Compliance | 5 | FCC Standard Certification | Medium |
| Logistics Cost | 3 | Bulk Liquid Tankers | High |
| Storage Stability | 6 | Climate-Controlled Warehousing | Medium |
The primary difference is concentration. Glacial ch3cooh is nearly 100% pure (anhydrous), whereas standard acetic acid is typically diluted with water (e.g., vinegar is about 5-8%). The "glacial" form is used in industrial and laboratory settings where water would interfere with the chemical reaction or where high potency is required for efficiency.
No, glacial ch3cooh is highly concentrated and corrosive; it can cause severe chemical burns to the skin, eyes, and digestive tract if ingested directly. It must be properly diluted according to strict safety guidelines and food-grade formulations before it can be used as an ingredient in consumer products.
It should be stored in containers made of 316L stainless steel or high-density polyethylene (HDPE) to prevent corrosion. The storage area should be cool, well-ventilated, and away from oxidizing agents. Because it freezes at 16.6°C, temperature-controlled environments are recommended to keep the product in liquid form for easier handling.
Yes, bio-based glacial ch3cooh produced via fermentation is chemically identical to the petroleum-based version. The only difference is the carbon source. Switching to bio-based versions helps companies reduce their carbon footprint and meet sustainability certifications without altering the performance or quality of their final food products.
Low-quality batches often exhibit a slight yellowish tint instead of being colorless, or they may contain trace amounts of heavy metals and organic impurities. These contaminants can lead to off-flavors in food products or trigger instability in pharmaceutical formulations, highlighting the importance of sourcing from certified food-grade suppliers.
By significantly lowering the pH level of a food product, glacial ch3cooh creates an acidic environment that is inhospitable to most pathogenic bacteria and spoilage molds. This inhibits enzymatic activity and microbial growth, effectively extending the shelf life of the product while maintaining its sensory characteristics.
In summary, glacial ch3cooh is an indispensable asset in the food manufacturing and chemical synthesis sectors, offering unparalleled purity and efficiency. From its role as a critical pH regulator to its potential as a sustainable, bio-based additive, its ability to enhance food safety and product stability is unmatched. By focusing on rigorous quality control and modern safety protocols, manufacturers can fully unlock the commercial value of this potent compound.
As the industry moves toward a greener and more digital future, the adoption of high-purity, sustainably sourced glacial ch3cooh will be a key differentiator for brands seeking to lead in quality and innovation. We encourage food manufacturers and chemical engineers to prioritize certified food-grade sources to ensure global compliance and consumer safety. Visit our website for more information: www.ysxlglacialaceticacid.com